CPWR Technical Report: Manganese Exposure and Cardiovascular Responses among Welders
Summary Statement
This study investigated the cardiac effects of Manganese exposures among welders, specifically boilermaker construction workers, using a repeated measures study design where workers were monitored twice or more over a 6-month period.
July 2010
Abstract
Welders are exposed to relatively high concentrations of manganese (Mn) and while the neurotoxicity of Mn is well documented, few studies have investigated the cardiac effects of occupational Mn exposures. The goal of this study was to investigate the cardiac effects of Mn exposures among welders, specifically boilermaker construction workers, using a repeated measures study design where workers were monitored twice or more over a 6-month period. A total of 78 male welders were recruited from a union hall out of Quincy, MA. In total, 118 workerdays were collected with 28 (36%) workers monitored at two or more time points. Blood, hair, and nail samples were collected as biomarkers of Mn exposure (Table 1). Hair samples were only available on 33% of participants due to short hair. Detailed work histories were collected to calculate a Mn cumulative exposure index (CEI). The cardiovascular health of each worker was assessed by ambulatory electrocardiogram (ECG) and inflammatory markers. A sub-study of 49 welders with toenail, blood and work history collected at one time-point was used to determine the relationship between blood and toenail Mn and CEI. Collected toenail samples had a skewed distribution ranging from 0.05 to 10.41 ng/g with a median of 0.80 ng/g (25th-75th percentiles: 0.45-1.49) (Table 2). Toenail Mn increased with the Mn-CEIs for the exposure windows between 7 and 12 months prior to the toenail clipping date. These results were confirmed when the Spearman correlations between toenail Mn and past Mn-CEI, adjusted for age and dietary Mn intake, were evaluated (Table 3). Biomarker and cardiovascular health endpoints analysis is ongoing. We hypothesize that occupational exposure to Mn will be associated with detectable changes in cardiovascular autonomic control as assessed by 24-hr heart rate variability (HRV) as well as changes in inflammatory markers as assessed by C - reactive protein and fibrinogen.
The top findings from this pilot study are:
- Toenails provide a useful biomarker of intermediate Mn exposures. Our study demonstrates that toenail Mn, averaged over clippings from all 10 toes, is correlated with Mn-CEI windows encompassing months 7-12 before the toenail clipping date.
- Toenail and blood Mn concentration showed no correlation and likely represent different exposure windows.
- Boilermaker construction workers with variable Mn exposure have detectable blood and toenail manganese concentrations.
- Toenail and blood samples are easily provided by welders while hair samples are difficult to collect.
- We were able to recruit 78 individuals into our study over the 18-month period. However, due to variable work schedules, it was difficult to monitor participants more than once (only 36%). Future studies should account for the variable work schedule among boilermaker construction workers.
Table 1: Summary of samples collected
| N (%) | |
| Metal Biomarker Sample | |
| Toenail | 94 (80) |
| Hair | 40 (33) |
| Blood | 105(89) |
| Cardiovascular Parameters | |
| ECG Holter Recordings | 87 (74) |
| Inflammatory Blood Markers | 110(93) |
Table 2. Distributions of toenail Mn by different characteristics of the participants and their cumulative exposure index levels (Mn-CEI) over different windows.
| Characteristics | Toenail Mn, ng/g | |||
| n | Mean (SD) | Median | 25th-75th Percentiles | |
| Mn-CEI | ||||
| For months 1-6 | ||||
|
19 | 0.17(0.24) | 0.79 | 0.27-1.49 |
|
14 | 1.75(0.98) | 0.93 | 0.32-1.63 |
|
16 | 7.92 (4.30) | 0.76 | 0.54-1.21 |
| For months 7-9 | ||||
|
16 | 0.06 (0.09) | 0.39 | 0.19-0.93 |
|
16 | 0.52 (0.20) | 0.84 | 0.57-1.49 |
|
17 | 2.28(1.03) | 1.10 | 0.51-1.70 |
| For months 10-12 | ||||
|
18 | 0.05 (0.07) | 0.58 | 0.27-0.81 |
|
18 | 0.57 (0.24) | 0.98 | 0.40-1.49 |
|
13 | 2.63(1.16) | 1.19 | 0.56-2.13 |
| For months 7-12 | ||||
|
19 | 0.20 (0.26) | 0.46 | 0.21-0.81 |
|
15 | 1.29(0.36) | 1.07 | 0.54-1.59 |
|
15 | 4.61 (2.12) | 1.10 | 0.54-1.70 |
| For past 12 months | ||||
|
20 | 0.64 (0.76) | 0.62 | 0.21-1.37 |
|
14 | 3.99(1.42) | 1.17 | 0.53-1.76 |
|
15 | 11.86(4.84) | 0.80 | 0.53-1.23 |
Table 3. Associations as assessed by partial spearman correlation coefficients between Toenail Mn (ng/g), blood Mn (ng/mL) and Mn-CEI (mg/m3-hr) exposure windows.
| Toenail Mn, ng/g | Blood Mn, ng/ml | ||||
| £ | P-value | £ | P-value | ||
| (n = 21) | |||||
| Blood Mn, ng/ml | 0.11 | 0.66 | 1.00 | ||
| Mn-CEI, mg/m3-hr | (n = 49) | (n = 27) | |||
| - Months 1-6 | 0.07 | 0.62 | 0.05 | 0.82 | |
| - Months 7-9 | 0.35 | 0.016* | 0.12 | 0.58 | |
| - Months 10-12 | 0.32 | 0.031* | 0.17 | 0.42 | |
| - Months 7-12 | 0.32 | 0.027* | 0.07 | 0.75 | |
| - Past 12 months | 0.19 | 0.19 | 0.15 | 0.46 | |
* Adjusted for age (year) and dietary Mn (mg/month).
Introduction
Welders are exposed to relatively high concentrations of manganese during the process of welding. Inhalation exposure occurs when molten mild or stainless steel, or manganesecontaining electrodes is volatilized. While the neurotoxicity of manganese exposure and the neurological health effects among welders are well documented (Sjogren, Iregren et al. 1996; Bowler, Gysens et al. 2003; Bowler, Gysens et al. 2006; Bowler, Roels et al. 2007), few studies have investigated the cardiac effects of occupational manganese exposures, despite the fact that welders are at increased risk for ischemic heart disease (Newhouse, Oakes et al. 1985; Moulin, Wild et al. 1993; Sjogren, Fossum et al. 2002).
The goal of this study was to investigate the cardiac effects of manganese exposures among welders using a repeated measures study design with two assessment points. Welders, specifically boilermaker construction workers, were recruited from a union hall out of Quincy, MA. A number of biomarkers of Mn exposure were collected including blood, hair, and nail. In addition, detailed work histories were also collected.
Cardiovascular health of each worker was assessed by ambulatory electrocardiogram. We expanded the original aims of the study by monitoring inflammatory markers as an additional cardiovascular health endpoint. We hypothesized that occupational exposure to manganese is associated with detectable changes in cardiovascular autonomic control as assessed by 24-hr heart rate variability (HRV) as well as changes in inflammatory markers as assessed by C - reactive protein and fibrinogen.
The original study objectives were to:
1. To quantify cumulative manganese exposure among boilermaker construction workers occupationally exposed to manganese-rich welding fume using cumulative exposure index (CEI) from work history and biomarkers of manganese exposure including blood, hair and nails and to identify the relationship between CEI and biomarkers of manganese exposure.
2. To conduct a prospective, repeated measures study of cardiovascular autonomic control as assessed by 24-hr HRV
3. To investigate the exposure-response relationship between cumulative manganese exposures (assessed by CEI, blood, hair and nail manganese) and 24-hr HRV.
The study objectives were expanded:
4. To investigate the exposure-response relationship between cumulative manganese exposures (assessed by CEI, blood, hair and nail manganese) and inflammatory markers including C - reactive protein and fibrinogen.
Background and Significance
Welders and manganese exposure
Welders are exposed to manganese during the process of welding when molten metal from mild or stainless steel or from manganese containing electrodes or wires is volatilized. Volatilized welding fume is small in size, mainly spherical particles ranging from 50 to 300 nm in diameter (Zimmer 2002). These small particles have the ability to reach deep into the alveolar region of the lung where they can initiate health effects. Alternately, toxicological studies suggest that the small particles may be transported via the olfactory nerves where they can initiate a cascade of central nervous system effects (Doty 2008).
Short-term, cross-shift exposures to occupational manganese exposure are best captured using personal sampling. The appropriate quantification of longer-term manganese exposures is less clear and can be quantified by a number of techniques including calculating a cumulative exposure index (CEI) of manganese dose based on detailed work histories and previous personal air manganese measurements or through use biomarkers of manganese exposure which provide cumulative internal manganese dose.
Biomarkers of manganese exposure
Numerous biological media including whole blood, serum, urine, nails, and hair have been investigated as potential biomarkers of manganese exposures. Monitoring occupational and environmental exposures to manganese is challenging because manganese is an essential element and concentrations of internal manganese are regulated by homeostatic mechanisms governing absorption, disposition and excretion. In general, the whole-body half-time of manganese is estimated to be about 37 days for healthy individuals without occupational exposures and anywhere from 15 to 28 days in healthy miners with or without chronic manganese poisoning (Michalke, Halbach et al. 2007).
Among workers with occupational manganese exposure, biomarkers of manganese exposure have been evaluated in welders (Ellingsen, Dubeikovskaya et al. 2006; Smith, Gwiazda et al. 2007), ferroalloy workers (Lucchini, Selis et al. 1995; Apostoli, Lucchini et al. 2000; Smith, Gwiazda et al. 2007) as well as manganese oxide and salt production workers (Roels, Lauwerys et al. 1987). In terms of acute manganese exposures, associations have been observed with workday air manganese and post-shift urine manganese (Apostoli, Lucchini et al. 2000; Ellingsen, Hetland et al. 2003). However, since over 95% of manganese is excreted via the bile to feces (Klaassen 1974), excretion in urine is low, making urine manganese a poor biomarker. Likewise, serum manganese concentrations have also been shown to be a poor biomarker as compared to manganese in whole blood (Smith, Gwiazda et al. 2007).
Both short-term (daily work shift) and long-term (months to years) manganese exposures have been related to whole blood manganese concentrations with mixed results. In terms of acute exposures, among the manganese oxide and salt workers, there was no relationship between workday air manganese concentrations and post-shift manganese in whole blood (Roels, Lauwerys et al. 1987). Yet, among the ferroalloy workers, a statistically significant linear exposure-response relationship was observed between work shift air manganese and post-shift whole blood manganese (Apostoli, Lucchini et al. 2000). Further evaluation of this cohort found heterogeneity in the linear association between whole blood manganese with consistent results observed in the low (median = 0.42 μg/m3) and medium (median = 4.2 μg/m3) exposure groups, but not in the high (median = 292 μg/m3) (Smith, Gwiazda et al. 2007). Among long-term exposed manganese oxide and salt workers (Roels, Lauwerys et al. 1987), dry cell battery workers (Bader, Dietz et al. 1999), and ferroalloy production workers (Apostoli, Lucchini et al. 2000) blood manganese concentrations were higher in manganese-exposed workers as compared to non-exposed control populations. Among bridge welders, a statistically significant association was observed among workers’ blood manganese concentration and CEI calculated over the previous 16 months (Smith, Gwiazda et al. 2007). Smith et al (2007) interpret the ferroalloy and bridge welder results to suggest that when manganese exposures are consistent, as is the case for the ferroalloy workers, manganese whole blood levels may represent average air manganese levels while blood manganese may reflect an integrative cumulative exposure when manganese exposures are highly variable or have been reduced, as is this case with the welders. This hypothesis is supported by a number of studies that have found elevated blood manganese levels to be associated with past cumulative exposure levels (Lucchini, Selis et al. 1995; Myers, Thompson et al. 2003). Both Apostoli (2000) and Lucchini (1995; 1999) suggest that blood manganese reflects the body burden of manganese in unexposed workers, and is a reflection of both body burden as well as acute exposures among exposed workers. Among ferroalloy workers who had temporarily stopped working (1 to 42 days), high correlations between blood manganese and CEIs were observed (Lucchini, Toffoletto et al. 1995; Lucchini, Bergamaschi et al. 1997). It is unclear what role individual characteristics play in the relationship between air manganese exposures and manganese body burden. Despite the uncertainty in biomarkers of manganese exposures, exposure-response relationships between whole blood manganese and a number of neurological outcomes have been found among welders (Bowler, Roels et al. 2007), suggesting that blood manganese may be an important exposure marker.
As compared to blood manganese, hair and nail manganese concentrations have yet to be explored as biomarkers of occupational manganese exposure. Both hair and nail samples have the advantage of being easy to collect and represent a longer time period of exposure as compared to blood manganese. Hair manganese represents a cumulative measure of manganese with the proximal 10 mm length sequence representing a measure of dose within the last 4 weeks and subsequent 10 mm lengths representing dose over the prior 4 weeks (Bader, Dietz et al. 1999). In a study in a dry cell battery manufacturing plant, manganese concentrations in hair taken from the arm pit were higher among exposed workers as compared to unexposed workers (Bader, Dietz et al. 1999). While no associations were observed between blood and hair manganese concentrations, an increase in hair manganese was observed with increased average manganese oxide exposures. Studies of environmental manganese exposures have found hair manganese to be an effective biomarker: chronic manganese exposure was associated with high hair manganese in a Greek population exposed via drinking water (Kondakis, Makris et al. 1989), low intelligence scores were associated with high hair manganese in children residing near a hazardous waste site (Wright, Amarasiriwardena et al. 2006), associations between hair manganese and drinking water manganese and also hyperactivity were observed among a small cohort of Canadian children (Bouchard, Laforest et al. 2007). Nail clippings also present a cumulative exposure measure as clippings from all 10 toes are likely to reflect exposure integrated over the previous 3 – 12 months time (Longnecker, Stampfer et al. 1993). However, only a few studies have evaluated the use of nail manganese as a biomarker (Wongwit, Kaewkungwal et al. 2004; Mehra and Juneja 2005). In a study of trace elements in fingernails of male workers, as compared to controls, higher nail manganese exposures were observed among participants reporting a number health problems including hypertension as well as skin disease, mental stress, diabetes, respiratory trouble, and ophthalmic disease (Mehra and Juneja 2005). Overall, studies have shown that manganese exposures deposit in both hair and nails.
Cardiovascular effects of manganese exposures
While the neurotoxicity of manganese exposure and the neurological health effects among welders is well documented (Sjogren, Iregren et al. 1996; Bowler, Gysens et al. 2003; Bowler, Gysens et al. 2006; Bowler, Roels et al. 2007), few studies have investigated the cardiac effects of occupational manganese exposures (Barrington, Angle et al. 1998; Jiang and Zheng 2005; Cavallari, Eisen et al. 2008), despite the fact that welders are at increased risk for ischemic heart disease (Newhouse, Oakes et al. 1985; Moulin, Wild et al. 1993; Sjogren, Fossum et al. 2002).
A small number of epidemiological studies have linked long- and short-term manganese exposures to cardiac effects, specifically looking at changes in cardiovascular autonomic control by examining electrocardiograms (ECG) for changes in heart rhythm including sinus arrhythmia and heart rate variability (HRV). Jiang and Zheng (2005) observed abnormal ECG parameters including sinus arrhythmia and ST-T changes in workers exposed to manganese oxide as compared to unexposed workers. Among manganese alloy workers, Barrington et al. (1998) report decreased autonomic function and low 24-hr HRV, a risk factor for future cardiovascular events (Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology 1996). Our recent research found a link between short-term exposure to manganese and changes in HRV, specifically we found a statistically significant exposure-response relationship between workday manganese exposures and declines in nocturnal HRV among boilermaker construction workers occupationally exposed to welding fume (Cavallari, Eisen et al. 2008).
It is biologically plausible that a relationship exists between manganese and changes in cardiovascular autonomic control. Given the regulation of the cardiovascular system by the autonomic nervous system, the cardiotoxic effects of manganese may be indirectly derived from the manganese effect on the autonomic nervous system. Furthermore, manganese has the ability to generate reactive oxygen species (Michalke, Halbach et al. 2007) which may initiate an inflammatory cascade which goes on to cause cardiovascular effects or in the case of neurological effects may promote the auto-oxidation and inactivation of dopamine (Hudson, Evans et al. 2001). In addition to causing an acute effect, it is likely that the long- and intermediate-term manganese exposures accumulate to cause changes in overall cardiovascular health.
This investigation was designed to evaluate the potential cardiotoxicity of intermediate-term manganese exposure. The study used heart rate variability (HRV) as a measure of cardiovascular autonomic control. HRV has the advantage of being easily obtained through the use of the noninvasive ambulatory electrocardiogram (ECG). In general, low HRV is a predictor of cardiac and all-cause mortality in the general population (Kors, Swenne et al. 2007). Since the heart is under constantly responding to the external environment, we propose the use of 24-hr HRV to give a measure of overall cardiovascular health without influence from ventilation or physical activity (Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology 1996). The study used fibrinogen and C-reactive protein (CRP) as markers of inflammation. CRP has been found to be a useful inflammatory biomarker of cardiovascular risk. In fact among healthy populations, elevated CRP is associated with increased risk of future myocardial infarction, strove, peripheral vascular disease and cardiovascular death (Blake and Ridker 2002).
Methods
Study design
A repeated measures study design was used (Table 1) to determine how changes in cardiovascular autonomic control and inflammation relate to manganese exposures. A major strength of this study design is that rather than compare workers to each other, each worker is compared to himself by examining the association between HRV and manganese exposures at two points in time, 6-months apart.
Table 1. Sampling Scheme
| Baseline | 6 months | |
| HRV | X | X |
| Questionnaire | X | X |
| Toenail Collection | ||
| Hair Collection | ||
| Blood Collection* | X | X |
Participants were recruited from a local boilermaker union, in Quincy, MA. Boilermakers are construction workers trained to weld upon round vessels, often located within power plants. Due to rotating energy needs throughout the year, boilermakers’ work is cyclic with intense periods of work and welding exposures occurring over a 5-6 month time period, followed by a down period when workers are laid off for up to 3-months time and have little or no welding exposures. Our original goal was to monitor workers during the no exposure period (when workers had been laid-off and were free from welding exposures for a minimum of 6-weeks) and then a second time at the end of the peak exposure period, approximately 6-months later. However, due to time constraints and availability of participants, workers were monitored at their convenience without regard to previous exposure. We believe that this change in the protocol will still allow us to evaluate the original study aims.
To eliminate the acute effects of welding, monitoring occurred on a day when participants were not exposed to welding fumes. Participants were fitted with an ambulatory ECG at the Boilermaker Union Hall and were asked to return 24-hrs later to return the ECG equipment.
At baseline and subsequent monitoring periods a self-administered questionnaire was used to collect medical history and medication use. Demographic and lifestyle information including smoking history and occupational history were also collected.
Physiologic and Biological Measurements
Holter monitoring
On each monitoring day, participants were fitted with a standard 5-lead ECG Holter monitor. To facilitate good lead contacts, the participant’s skin was shaved, if necessary, cleansed with an alcohol wipe, and slightly abraded. Participants were asked to go about their normal nonworkday activities while refraining from welding fume exposures. Participants were asked to keep a diary of activities including quantity of cigarettes and second-hand smoke exposure, quantity of caffeine and alcohol consumption and sleep duration. Each 24-hr tape is being analyzed by the Cardiovascular Epidemiology Group of the Beth Israel Deaconess Medical Center, Boston, Ma led by Dr. Murray Mittleman.. Tapes will be analyzed in the time domain, and indices including the square root of the mean of the sum of the squared differences between adjacent NN intervals (rMSSD), the standard deviation of all NN intervals over the entire period (SDNN), and the mean of the standard deviations of all NN intervals for all 5-min segments (SDNNi) will be calculated over the entire 24-hr recording.
Inflammatory marker assessment
Participants were asked to provide one blood sample. Blood samples were drawn by a phlebotomist using standard clinical procedures. Blood samples were analyzed by the Laboratory Corporation of America (LabCorp), a CLIA-certified clinical laboratory that operates under standard operating procedures with a prompt turnaround time. The blood will also be analyzed for fibrinogen, a marker of platelet activation and C-reactive protein (CRP) a marker of inflammation and predictor of coronary heart disease. In addition, a standard lipid panel was performed.
Manganese exposure assessment
Bioaccumulation of manganese exposures was assessed by evaluation of manganese concentrations in blood, hair and nails. We hypothesize that each of the different biological media present a different window of exposure; blood a 1-month exposure window, hair a 1 – 3 month exposure window, and nails a 9-12 month exposure window. At baseline and 6 months, a whole blood sample will be drawn. Whole blood samples were collected by a trained and certified professional by venipuncture using a butterfly catheter and heparinised tubes. The first 2 mL of each blood sample was rejected due to possible contamination from the steel needle. Participants were asked to provide toenail clippings. When available, hair samples were taken from the occipital region using stainless steel scissors.
The biological samples were analyzed in the Harvard School of Public Health Trace Metals Laboratory, which is equipped with a class 100 clean room facility and a dynamic reaction cellinductively coupled plasma mass spectrometer (DRC-ICP-MS, Elan 6100, Perkin Elmer, Norwalk, CT). Quality control measures are routinely performed in the laboratory and include analysis of initial calibration verification standards (NIST SRM 1643d trace elements in water), continuous calibration standards, procedural blanks, duplicate samples, spiked samples, QC standard, and certified reference material.
Toenail clippings were prepared and analyzed using standard methods (Kile, Houseman et al. 2007). Briefly, external contamination was removed from nails by sonicating samples in a 1% Triton X-100 solution (Sigma-Aldrich, Inc., St. Louis, MO) for 20 minutes. Toenails were then rinsed repeatedly in Milli-Q water (Millipore Corp., Billerica, MA), dried, weighed, digested in nitric acid, and analyzed using the ICP-MS. Each sample was subjected to five replicate analyses. The net averaged concentration was calculated by subtracting detectable laboratory blank concentrations within each batch.
Mn-CEIs for exposure windows of interest were calculated using each welder’s report of specific welding tasks and total hours of welding for that task during the given exposure window, and the reported use of respiratory protection. Previous information on manganese content while welding at the apprentice hall (Cavallari, Eisen et al. 2008) and while at overhaul of boilers (Liu, Woodin et al. 2005) were used to assign manganese concentration to each task. The Mn-CEI for an exposure window in the past 12 months was derived by summing (equation 1) the products of the average Mn exposure intensity, the number of hours worked on that particular task, and an adjustment for the effect of respiratory protection used for each welding task the participant performed during that exposure window (equation 2).

Where MnAi is the average air Mn concentration for the ith task performed; Timewi is the total hours spent for the ith task performed in the wth exposure window; %respiratorwij is the estimated percentage of time the participant used the jth respirator in the ith task performed in the wth exposure window; and APFj is the assigned protection factor for the jth respirator used. This yields a measure of cumulative exposure across all exposed jobs in that exposure window, in mg/m3-hr.
Results
Data entry, analysis of biomarker metal concentrations, and analysis of HRV is on-going. The available summary data is presented. Data analysis and interpretation is pending completion of sample analysis and data entry.
A total of 78 boilermaker construction workers were recruited from the Boilermakers Union Local 29 in Quincy, MA for participation in the study. Monitoring workers repeatedly over the short time period proved difficult. Twenty-eight (36%) workers were monitored more than once with 18 (23%) workers monitored on two occasions, 8 (10%) workers on three occasions, and 2 (3%) workers monitored on four occasions. Background characteristics of a portion of the participants are provided in Table 2. A summary of the samples collected is presented in Table 3. Unfortunately, due to short-hair and crew cuts, collection of hair proved difficult. Only 40 hair samples were collected and due to small sample size were not analyzed for Mn.
Table 2: Participant Characteristics for 36 of the 78 participants
| Characteristic | N (%) or Mean (SD) |
| Male | 36 (100) |
| Age* | 38 (10) |
| Smokers** | 15 (48) |
| Race | |
| White | 29 (81) |
| Black | 6 (17) |
| Hispanic | 1 (2) |
*Data available for 28 participants, pending for remaining
**Data available for 31 participants, pending for remaining
Table 3: Summary of samples collected
| N (%) | |
| Metal Biomarker Sample | |
| Toenail | 94 (80) |
| Hair | 40 (33) |
| Blood | 105 (89) |
| Cardiovascular Parameters | |
| ECG Holter Recordings | 87 (74) |
| Inflammatory Blood Markers | 110 (93) |
Sample analysis was performed in-house by the Trace Metals Laboratory of Harvard School of Public Health. The cost savings associated with performing the analysis in-house, allowed us to analyze the samples for Pb, Cd, and Ni in addition to Mn. Preliminary results are presented below (Table 4). As previously noted, we expanded our cardiovascular endpoints to include markers of inflammation. Preliminary summary measures are presented (Table 5).
Table 4: Summary of biomarkers of Mn and other metals in 20 boilermaker welders
| N | Mean | Median | Std Dev |
Min | - | Max | ||
| Toenail (μg/g) | Mn | 15 | 0.66 | 0.51 | 0.45 | 0.21 | - | 2.01 |
| Pb | 15 | 0.59 | 0.23 | 0.87 | 0.12 | - | 3.52 | |
| Cd | 15 | 0.05 | 0.01 | 0.11 | 0.004 | - | 0.45 | |
| Ni | 15 | 5.26 | 1.94 | 7.24 | 0.12 | - | 23.29 | |
| Blood (μg/g) | Mn | 20 | 1.14 | 1.09 | 0.31 | 0.76 | - | 1.83 |
| Pb | 20 | 2.01 | 1.76 | 1.16 | 0.46 | - | 6.14 | |
| Cd | 20 | 0.06 | 0.04 | 0.10 | 0.01 | - | 0.44 | |
| Ni | 20 | 0.59 | 0.56 | 0.07 | 0.50 | - | 0.74 | |
| Urine (μg/g) | Mn | 20 | 2.17 | 2.17 | 0.70 | 0.80 | - | 3.43 |
| Pb | 20 | 0.98 | 0.67 | 1.02 | ND* | - | 4.32 | |
| Cd | 20 | 0.58 | 0.33 | 0.58 | ND** | - | 2.29 | |
| Ni | 20 | 11.43 | 9.50 | 5.68 | 1.32 | - | 23.73 | |
**n=3 below detection limit
Table 4: Summary of blood inflammatory markers and lipids
| N | Mean (SD) | |
| Inflammatory Markers | ||
| C-Reactive Proteins (mg/l) | 77 | 1.94 (2.08) |
| Fibrinogen (mg/L) | 40 | 232 (59) |
| Lipid Levels | ||
| Total Cholesterol (mg/dL) | 71 | 196 (45) |
| Triglycerides (mg/dL) | 71 | 169 (142) |
| HDL (mg/dL) | 71 | 44 (15) |
| LDL (mg/dL) | 67 | 117 (33) |
Evaluation of Biomarkers of Mn at one time-point
A sub-study of 46 welders with toenail, blood and work history collected at one time-point was used to determine the relationship between blood and toenail Mn and cumulative exposure index. Results of this sub-study of 46 welders are presented. Correlation coefficients, adjusted for age (year) and dietary Mn (mg/month), were computed to assess the associations between toenail Mn and Mn-CEI for the exposure windows of interest. Due to the log normal distribution, spearman correlations were used to assess the association between tow nail Mn and cumulative exposure index Statistical analyses were performed with Statistical Analysis System (SAS Institute Inc., NC) version 9.1.3. Statistical significance was set at alpha = 0.05.
Collected toenail samples (n=49) had a skewed distribution ranging from 0.05 to 10.41 ng/g with a median of 0.80 ng/g (25th-75th percentiles: 0.45-1.49) (Table 5). The distribution of toenail Mn by different characteristics and Mn-CEI for the different exposure windows, are presented in table 1. Toenail Mn tended to increase with age, was higher in whites, and increased with the Mn-CEIs for the exposure windows between 7 and 12 months prior to the toenail clipping date.
Table 5. Distributions of toenail Mn by different characteristics of the participants and their cumulative exposure index levels (Mn-CEI) over different windows.
| Characteristics | Toenail Mn, ng/g | |||
| n | Mean (SD) | Median | 25th – 75th Percentiles | |
| Age | ||||
| - < 35 years | 17 | 26.9 (4.6) | 0.46 | 0.26 – 1.49 |
| - 35 to 50 years | 15 | 41.1 (4.5) | 0.79 | 0.50 – 1.17 |
| - > 50 years | 17 | 55.5 (3.6) | 1.10 | 0.56 – 1.95 |
| Years of Welding | ||||
| - < 5 years | 19 | 1.66 (0.8) | 0.69 | 0.30 – 1.59 |
| - 5 to 10 years | 15 | 7.58 (1.8) | 0.80 | 0.54 – 1.13 |
| - > 10 years | 15 | 25.6 (7.1) | 0.81 | 0.35 – 1.61 |
| Race | ||||
| - White | 40 | 0.91 | 0.51 – 1.54 | |
| - Non-white | 9 | 0.46 | 0.11 – 0.46 | |
| Body Mass Index | ||||
| - <25 | 7 | 23.15 (1.0) | 0.56 | 0.30 – 1.40 |
| - 25-30 | 26 | 27.18 (1.2 | 0.81 | 0.45 – 1.26 |
| - >30 | 14 | 34.12 (3.1) | 0.74 | 0.46 – 1.44 |
| Dietary Mn | ||||
| - <40 mg/month | 15 | 25.55 (12.2) | 0.65 | 0.37 – 1.76 |
| - 40-85 mg/month | 17 | 58.57 (11.5) | 0.86 | 0.58 – 1.24 |
| - >85 mg/month | 17 | 145.4 (51.7) | 0.79 | 0.44 – 1.27 |
| Mn-CEI | ||||
| For months 1–6 | ||||
| - < 0.7 mg/m3-hr | 19 | 0.17 (0.24) | 0.79 | 0.27 – 1.49 |
| - 0.7 – 4.0 mg/m3-hr | 14 | 1.75 (0.98) | 0.93 | 0.32 – 1.63 |
| - >4.0 mg/m3-hr | 16 | 7.92 (4.30) | 0.76 | 0.54 – 1.21 |
| For months 7–9 | ||||
| - < 0.3 mg/m3-hr | 16 | 0.06 (0.09) | 0.39 | 0.19 – 0.93 |
| - 0.3 – 1.0 mg/m3-hr | 16 | 0.52 (0.20) | 0.84 | 0.57 – 1.49 |
| - >1.0 mg/m3-hr | 17 | 2.28 (1.03) | 1.10 | 0.51 – 1.70 |
| For months 10–12 | ||||
| - < 0.3 mg/m3-hr | 18 | 0.05 (0.07) | 0.58 | 0.27 – 0.81 |
| - 0.3 – 1.0 mg/m3-hr | 18 | 0.57 (0.24) | 0.98 | 0.40 – 1.49 |
| - >1.0 mg/m3-hr | 13 | 2.63 (1.16) | 1.19 | 0.56 – 2.13 |
| For months 7–12 | ||||
| - < 0.7 mg/m3-hr | 19 | 0.20 (0.26) | 0.46 | 0.21 – 0.81 |
| - 0.7 – 2.0 mg/m3-hr | 15 | 1.29 (0.36) | 1.07 | 0.54 – 1.59 |
| - >2.0 mg/m3-hr | 15 | 4.61 (2.12) | 1.10 | 0.54 – 1.70 |
| For past 12 months | ||||
| - < 1.5 mg/m3-hr | 20 | 0.64 (0.76) | 0.62 | 0.21 – 1.37 |
| - 1.5 – 7.0 mg/m3-hr | 14 | 3.99 (1.42) | 1.17 | 0.53 – 1.76 |
| - >7.0 mg/m3-hr | 15 | 11.86 (4.84) | 0.80 | 0.53 – 1.23 |
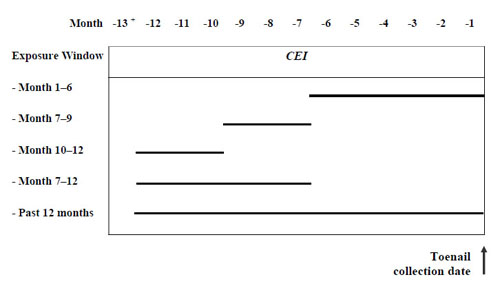
The Spearman correlations between toenail Mn and past Mn-CEI, adjusted for age and dietary Mn intake, were significant for the 3 Mn-CEI windows encompassing months 7-12 before the toenail clipping date (7th-9th months, 10th-12th months, and 7th-12th months), ranging from 0.32 to 0.35 (Table 6). The unadjusted correlations were similar but slightly weaker. Pearson correlation analyses on square root transformed toenail Mn and Mn-CEI also yielded similar coefficients. There was little correlation between toenail Mn and any of the other Mn-CEI exposure. Our study demonstrates that toenail Mn, averaged over clippings from all 10 toes, is correlated with Mn-CEI windows encompassing months 7-12 before the toenail clipping date, which is the exposure window it would be expected to reflect based on toenail growth rates. (Figure 1)
Table 6. Associations as assessed by partial spearman correlation coefficients between Toenail
| Mn (ng/g), blood Mn (ng/mL) and Mn-CEI (mg/m3-hr) exposure windows. | ||||
| Toenail Mn, ng/g | Blood Mn, ng/ml | |||
| ρ | P-value | ρ | P-value | |
| (n = 21) | ||||
| Blood Mn, ng/ml | 0.11 | 0.66 | 1.00 | |
| Mn-CEI, mg/m3-hr | (n = 49) | (n = 27) | ||
| - Months 1–6 | 0.07 | 0.62 | 0.05 | 0.82 |
| - Months 7–9 | 0.35 | 0.016* | 0.12 | 0.58 |
| - Months 10–12 | 0.32 | 0.031* | 0.17 | 0.42 |
| - Months 7–12 | 0.32 | 0.027* | 0.07 | 0.75 |
| - Past 12 months | 0.19 | 0.19 | 0.15 | 0.46 |
Figure 1. Exposure windows of interest for Mn-CEI and toenail Mn.
Summary
We have surpassed the original aims of the small study project. Our original proposal projected 40 participants over 80 worker-days. We have monitored 78 workers over 118 worker-days. In addition to examining cardiovascular autonomic health using HRV, we were also able to monitor inflammatory markers an additional independent predictor of cardiovascular disease. Some changes to the original study design were made. Due to participant availability, we were unable to monitor each participant twice over a 6-month period. However, we believe we have sufficient sample size to examine the relationship of interest. We were also unable to collect hair samples on the majority of the participants. However, preliminary results suggest that toenails may provide a useful biomarker of intermediate Mn exposures.
Due to additional sampling time periods, sample analysis was delayed and is on-going. Evaluation of the relationship between cumulative Mn and cardiovascular parameters will occur once all data has been received.
This small project has already provided information on biomarkers of manganese exposure among welders intermittently exposed to manganese-rich welding fume (which is in a manuscript that is currently in review). We are looking forward to examining the association of biomarkers of manganese exposure and cumulative exposure index repeatedly over a 6-month time period. By examining the same individual over time we will be able to control for individual difference in metal metabolism and have a clearer signal of the relationship between intermediate Mn exposure and toenail and blood Mn. Likewise, we look forward to investigating the cardiac effects of intermediate exposures to manganese among welders.
Bibliography
Apostoli, P., R. Lucchini, et al. (2000). "Are current biomarkers suitable for the assessment of manganese exposure in individual workers?" Am J Ind Med 37(3): 283-90.
Bader, M., M. C. Dietz, et al. (1999). "Biomonitoring of manganese in blood, urine and axillary hair following low-dose exposure during the manufacture of dry cell batteries." Int Arch Occup Environ Health 72(8): 521-7.
Barrington, W. W., C. R. Angle, et al. (1998). "Autonomic function in manganese alloy workers." Environ Res 78(1): 50-8.
Blake, G. J. and P. M. Ridker (2002). "Inflammatory bio-markers and cardiovascular risk prediction." J Intern Med 252(4): 283-94.
Bouchard, M., F. Laforest, et al. (2007). "Hair manganese and hyperactive behaviors: pilot study of school-age children exposed through tap water." Environ Health Perspect 115(1): 122- 7.
Bowler, R. M., S. Gysens, et al. (2003). "Neuropsychological sequelae of exposure to welding fumes in a group of occupationally exposed men." Int J Hyg Environ Health 206(6): 517- 29.
Bowler, R. M., S. Gysens, et al. (2006). "Manganese exposure: neuropsychological and neurological symptoms and effects in welders." Neurotoxicology 27(3): 315-26.
Bowler, R. M., H. A. Roels, et al. (2007). "Dose-effect relationships between manganese exposure and neurological, neuropsychological and pulmonary function in confined space bridge welders." Occup Environ Med 64(3): 167-77.
Cavallari, J. M., E. A. Eisen, et al. (2008). "PM2.5 metal exposures and nocturnal heart rate variability: a panel study of boilermaker construction workers." Environ Health 7: 36.
Doty, R. L. (2008). "The olfactory vector hypothesis of neurodegenerative disease: is it viable?" Ann Neurol 63(1): 7-15.
Ellingsen, D. G., L. Dubeikovskaya, et al. (2006). "Air exposure assessment and biological monitoring of manganese and other major welding fume components in welders." J Environ Monit 8(10): 1078-86.
Ellingsen, D. G., S. M. Hetland, et al. (2003). "Manganese air exposure assessment and biological monitoring in the manganese alloy production industry." J Environ Monit 5(1): 84-90.
Hudson, N. J., A. T. Evans, et al. (2001). "Effect of process parameters upon the dopamine and lipid peroxidation activity of selected MIG welding fumes as a marker of potential neurotoxicity." Ann Occup Hyg 45(3): 187-92.
Jiang, Y. and W. Zheng (2005). "Cardiovascular toxicities upon manganese exposure." Cardiovasc Toxicol 5(4): 345-54.
Kile, M. L., E. A. Houseman, et al. (2007). "Association between total ingested arsenic and toenail arsenic concentrations." J Environ Sci Health A Tox Hazard Subst Environ Eng 42(12): 1827-34.
Klaassen, C. D. (1974). "Biliary excretion of manganese in rats, rabbits, and dogs." Toxicol Appl Pharmacol 29(3): 458-68.
Kondakis, X. G., N. Makris, et al. (1989). "Possible health effects of high manganese concentration in drinking water." Arch Environ Health 44(3): 175-8.
Kors, J. A., C. A. Swenne, et al. (2007). "Cardiovascular disease, risk factors, and heart rate variability in the general population." J Electrocardiol 40(1 Suppl): S19-21.
Liu, Y., M. A. Woodin, et al. (2005). "Estimation of personal exposures to particulate matter and metals in boiler overhaul work." J Occup Environ Med 47(1): 68-78.
Longnecker, M. P., M. J. Stampfer, et al. (1993). "A 1-y trial of the effect of high-selenium bread on selenium concentrations in blood and toenails." Am J Clin Nutr 57(3): 408-13.
Lucchini, R., P. Apostoli, et al. (1999). "Long-term exposure to "low levels" of manganese oxides and neurofunctional changes in ferroalloy workers." Neurotoxicology 20(2-3): 287-97.
Lucchini, R., E. Bergamaschi, et al. (1997). "Motor function, olfactory threshold, and hematological indices in manganese-exposed ferroalloy workers." Environ Res 73(1-2): 175-80.
Lucchini, R., L. Selis, et al. (1995). "Neurobehavioral effects of manganese in workers from a ferroalloy plant after temporary cessation of exposure." Scand J Work Environ Health 21(2): 143-9.
Lucchini, R., F. Toffoletto, et al. (1995). "Neurobehavioral functions in operating theatre personnel exposed to anesthetic gases." Med Lav 86(1): 27-33.
Mehra, R. and M. Juneja (2005). "Fingernails as biological indices of metal exposure." J Biosci 30(2): 253-7.
Michalke, B., S. Halbach, et al. (2007). "Speciation and toxicological relevance of manganese in humans." J Environ Monit 9(7): 650-6.
Moulin, J. J., P. Wild, et al. (1993). "A mortality study among mild steel and stainless steel welders." Br J Ind Med 50(3): 234-43.
Myers, J. E., M. L. Thompson, et al. (2003). "The utility of biological monitoring for manganese in ferroalloy smelter workers in South Africa." Neurotoxicology 24(6): 875-83.
Newhouse, M. L., D. Oakes, et al. (1985). "Mortality of welders and other craftsmen at a shipyard in NE England." Br J Ind Med 42(6): 406-10.
Roels, H., R. Lauwerys, et al. (1987). "Relationship between external and internal parameters of exposure to manganese in workers from a manganese oxide and salt producing plant." Am J Ind Med 11(3): 297-305.
Sjogren, B., T. Fossum, et al. (2002). "Welding and ischemic heart disease." Int J Occup Environ Health 8(4): 309-11.
Sjogren, B., A. Iregren, et al. (1996). "Effects on the nervous system among welders exposed to aluminium and manganese." Occup Environ Med 53(1): 32-40.
Smith, D., R. Gwiazda, et al. (2007). "Biomarkers of Mn exposure in humans." Am J Ind Med 50(11): 801-11.
Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology (1996). "Heart rate variability: standards of measurement, physiological interpretation and clinical use." Circulation 93(5): 1043-65.
Wongwit, W., J. Kaewkungwal, et al. (2004). "Comparison of biological specimens for manganese determination among highly exposed welders." Southeast Asian J Trop Med Public Health 35(3): 764-9.
Wright, R. O., C. Amarasiriwardena, et al. (2006). "Neuropsychological correlates of hair arsenic, manganese, and cadmium levels in school-age children residing near a hazardous waste site." Neurotoxicology 27(2): 210-6.
Zimmer, A. T. (2002). "The influence of metallurgy on the formation of welding aerosols." J Environ Monit 4(5): 628-32.


